Description
The highly conserved gene fragment of ORF1ab and N of COVID-19 are selected as the target to design special primers and Taqman probes. The conserved gene fragment of ORF1ab and N will be amplified accurately by specific primers, and detected by the taqman probes. During the process of PCR amplification, the Taq enzyme will cut off the fluorophore (at the 5′ end) from the probe, so that the fluorophore can dissociate in the reaction system and get rid of the shielding of quencher (at the 3′ end). Then, the fluorophore will be stimulated by light and emit fluorescence which can be detected by instruments. In this way, COVID-19 nucleic acid can be detected in the fully automatic enclosed reaction system.The internal control in the kit was used a specific primer for a pair of housekeeping genes and specific probes. The probe can specifically combine with a segment of nucleic acid template in the region of primer amplification. The internal control selects HEX/VIC channel. Thus, the monitoring process in the closed reaction system can be realized and the occurrence of false negatives can be monitored effectively.
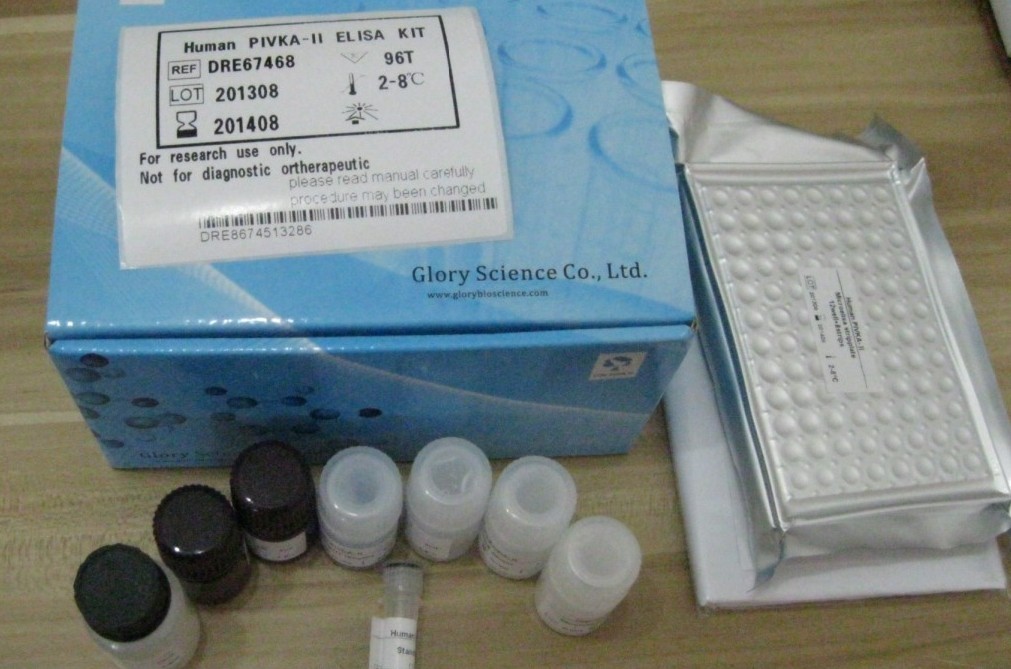
Kit Content:
Reagents Specification
CoV ORF1ab/N Reaction Solution 850μl×1
CoV ORF1ab/N Enzyme Mix 75μl×1
CoV ORF1ab/N Primer and Probe Mix 75μl×1
CoV ORF1ab/N Positive Control 200μl×1
Negative Control 200μl×1
Note: Do not mix the regents from different batches.
Specification:50T
Storage and validity:All reagents could be stored at -15℃~-25℃ for 12 months. The stability of unspent reagents would not be influenced by re-storage. But the thawing and freezing should not be more than three times. The opened reagents should be placed no more than 8 hours at room temperature. The products should be transport by ice box or refrigerated truck uner 2℃~8℃. Simulated transport tests indicate that the stability and validity could not be influenced by transport.
Instrument:Instruments with FAM, VIC (HEX), ROX/TEXAS RED channels, such as ABI, Bio-Rad and real time PCR systems.
Limitation:Test results cannot be directly used as the basis for clinical diagnosis or exclusion, only for reference by clinicians. At the same time, sample collection, preservation and improper transportation conditions, variation in the target sequence of influenza viruses or the sequence change caused by other causes could lead to a false negative result.
Properties:
1.Limit of detection(LOD): The LOD of the kit is 500 copies/ml.
2.Precision: Choose one high concentration and one low concentration as precision test sample. Repeat the test ten times. Variable coefficient of Ct value (CV, %) ≤ 5.0%.